Voltage-Gated Ion Channels in the Regulation of Cardiac Excitability
 Functional and Molecular Diversity of Native Cardiac Kv and Nav Channels
Functional and Molecular Diversity of Native Cardiac Kv and Nav Channels

Schematic of a cardiac Nav1.5 α subunit with transmembrane Navβ subunits, multiple intracellular accessory and interacting proteins.
Our efforts are focused on defining the physiological mechanisms that control the expression, properties and functioning of myocardial voltage-gated K+ (Kv) and Na+ (Nav) channels, which are key determinants of membrane excitability, affecting cardiac action potential waveforms and the propagation of electrical activity through the heart. Biochemical and mass spectrometry based proteomic approaches are employed to identify the components of native myocardial Kv and Nav channel complexes and electrophysiological and molecular strategies are combined in mouse models to define the functional significance of post-transcriptional Kv/Nav channel regulatory mechanisms.
Differential Expression of Kv Channels in Human Ventricular Myocytes.
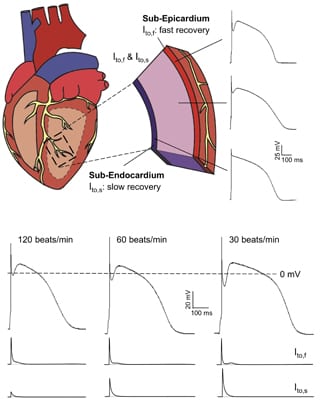
Rate-dependent contribution of Ito,s, but not Ito,f, to human LV action potential waveforms.
To investigate human heart electrical functioning directly, we developed robust methods for the isolation, in vitro maintenance, and electro-physiological characterization of Ca2+ tolerant, rod-shaped human left ventricular (LV) myocytes. In recent studies, we identified two transient Kv currents, Ito,f and Ito,s, with markedly different rates of recovery from inactivation in nonfailing human LV myocytes. Both Ito,f and Ito,s contribute to the action potential “notch”, particularly prominent in LV sub-epicardial myocytes (right). At high heart rates, Ito,s is markedly decreased, resulting in reductions in the notch potential and in action potential durations (right).
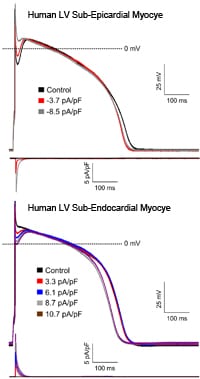
Effect on changing Ito,f on human LV action potential waveforms.
We also developed a mathematical model of Ito,f and used this in dynamic clamp experiments to manipulate Ito,f computationally in situ and in real time during current-clamp recordings. As illustrated in the figure (right), subtracting Ito,f in human LV sub-epicardial cells primarily attenuates the “notch”; action potential amplitudes and durations, however, are not measurably affected. In human LV sub-endocardial cells, addition of Ito,f produces a “notch” and shortens action potential durations.
Voltage-Gated Ion Channels in the Regulation of Neuronal Excitability
Another major focus in the lab is on defining the molecular and cellular mechanisms that control the diversity, expression, localization, properties and functioning of native neuronal Kv and Nav channels. These channels are key determinants of neuronal excitability, contributing to the control of resting potentials, action potential waveforms, repetitive firing patterns, responses to synaptic inputs and synaptic plasticity. We use in vivo and in vitro molecular genetic strategies, in combination with electrophysiological and behavioral approaches, to identify the molecular determinants of native neuronal Nav and Kv channels and to define the roles of these channels in shaping action potential waveforms, regulating repetitive firing, and controlling physiology and behavior.
Nav Channel Accessory Proteins Fine Tune Repetitive Firing in Cerebellar Purkinje Neurons
Similar to cardiac Nav channels, native neuronal Nav channels function in macromolecular protein complexes, comprising the pore-forming (α) subunit and multiple accessory subunits. In recent studies focused on defining the physiological roles of Nav channel accessory subunits, we found that the targeted deletion of intracellular fibroblast growth factor (iFGF14) markedly attenuates repetitive firing in cerebellar Purkinje neurons. Voltage-clamp experiments revealed that the disruption of firing in Fgf14-/- Purkinje neurons results from a hyperpolarizing shift in the voltage-dependence of steady-state inactivation of the transient Nav current component; loss of iFGF14 does not affect the persistent or the resurgent Nav current components. Spontaneous and evoked repetitive firing were also reduced in (Scn4b-/-) Purkinje neurons lacking the Nav beta 4 (Navβ4) accessory subunit. Whole-cell voltage-clamp recordings demonstrated that the resurgent Nav current was selectively reduced, but was not eliminated, in Scn4b-/- Purkinje neurons. Additional mechanisms, independent of Navβ4, therefore, contribute to the generation of resurgent Nav currents. Ongoing efforts are focused on identifying these mechanisms.
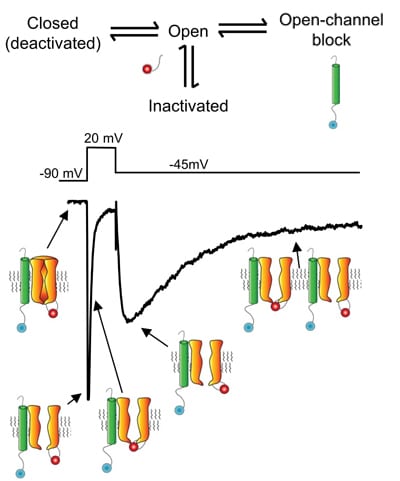
Schematic showing the waveforms of Nav currents in cerebellar Purkinje neurons and possible mechanisms contributing to the resurgent Nav current (evoked on membrane hyperpolarization to -40 mV from +25 mV). The intrinsic channel inactivation gate is shown in red and a putative open-channel blocking particle is depicted in blue. Ongoing experiments are focused on defining the roles of these (block vs. inactivation) mechanisms in the generation of resurgent Nav current.
Voltage-Gated Channels in the Regulation of Circadian Rhythms
The suprachiasmatic nucleus (SCN) is the master circadian pacemaker driving daily rhythms in mammalian physiology and behavior. SCN neurons utilize a transcription/translation feedback loop to generate daily changes in excitable membrane properties: SCN neurons fire repetitively at higher frequencies during the day than at night (see figure). Considerable evidence suggests roles for subthreshold K+ conductance(s) in mediating the day-night changes in repetitive firing rates, although the critical K+ conductance pathway(s) have not been identified. Our ongoing efforts are focused on identifying the molecular determinants of these K+ channels and delineating the mechanisms that couple these channels to the clock.
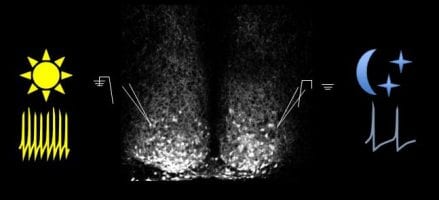
Schematic illustrating differences in daytime and nighttime repetitive firing rates in the SCN neurons.
Regulation of Ion Channel Expression and Remodeling
Heart failure, which afflicts more than five million adults in the United States alone, is associated with increased risk of ventricular arrhythmias and sudden cardiac death. The cellular, molecular and systemic mechanisms involved in the coordinated regulation of myocardial membrane excitability and in the maintenance of normal cardiac rhythms, as well as how these mechanisms go awry in the failing human heart, however, remain poorly understood. In studies focused on defining these mechanisms, we have found that long non-coding RNAs (lncRNAs) are differentially expressed, dynamically regulated in heart failure, and are more sensitive than messenger RNAs (mRNAs) or microRNAs (miRNAs) in discriminating cardiomyopathy phenotypes. Many of the lncRNAs are cardiac-enriched and show patterns of expression that are correlated with the expression levels of genes encoding transcription factors and ion channel subunits. Ongoing experiments are focused on exploring the roles of cardiac specific lncRNAs in the physiological regulation and pathophysiological remodeling of myocardial membrane excitability.